Biotherapeutics Manufacturing Centre
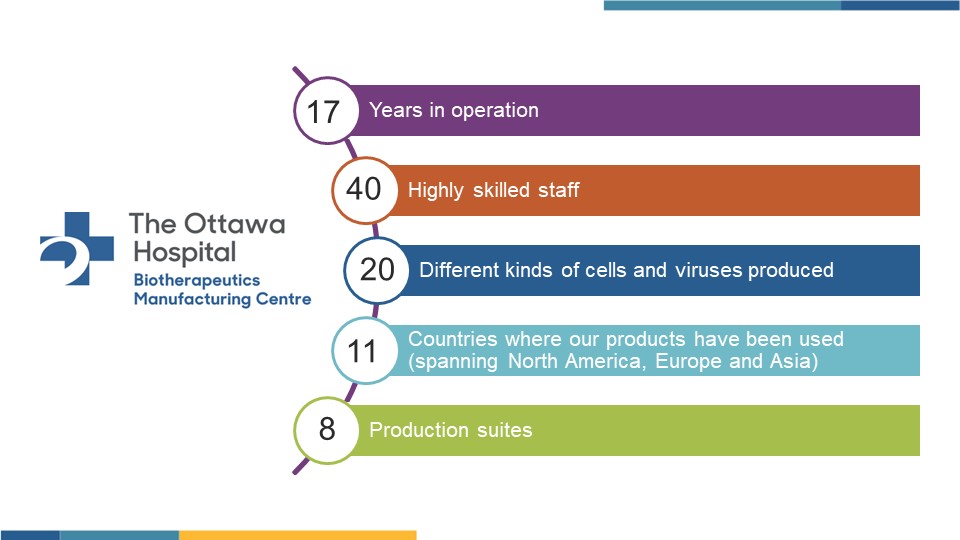
The Ottawa Hospital’s Biotherapeutics Manufacturing Centre (BMC) is the most experienced and successful facility of its kind in Canada, having manufactured more than 20 different cell and virus-based products for human clinical trials in Canada, the United States, Europe and Asia.
With eight manufacturing suites and 40 full-time staff, BMC offers a full range of services to both academic and corporate partners, including process development, manufacturing and fill-finish, all done according to Good Manufacturing Practice (GMP) standards.
Over the last 17 years, BMC has manufactured:
- Viral immunotherapies for cancer
- Mesenchymal stem / stromal cells
- CAR-T therapy (cells plus lentivirus)
- Genetically engineered endothelial progenitor cells
- Adeno Associated Viruses for gene therapy
- Vaccines for COVID-19
BMC also leads the only hands-on training program in Canada in biotherapeutics manufacturing, in partnership with Algonquin College, the University of Ottawa and Mitacs.
BMC has been supported by the Canada Foundation for Innovation, the Ontario Research Fund, BioCanRx (a Canadian Network of Centres of Excellence) and generous donors to The Ottawa Hospital Foundation.
“Finding a facility in Canada that could ready our DNA vaccine in vials time for the Phase 1 trials was extremely challenging because the few that we have here are all in high demand right now. We were thrilled to partner with The Ottawa Hospital’s Biotherapeutics Manufacturing Centre and benefit from their world-class expertise.”
John Lewis, CEO, Entos Pharmaceuticals
